The microbiome, denoting the harmonious assembly of microorganisms within a host, is essential for the optimal operation of plants and animals, including humans.
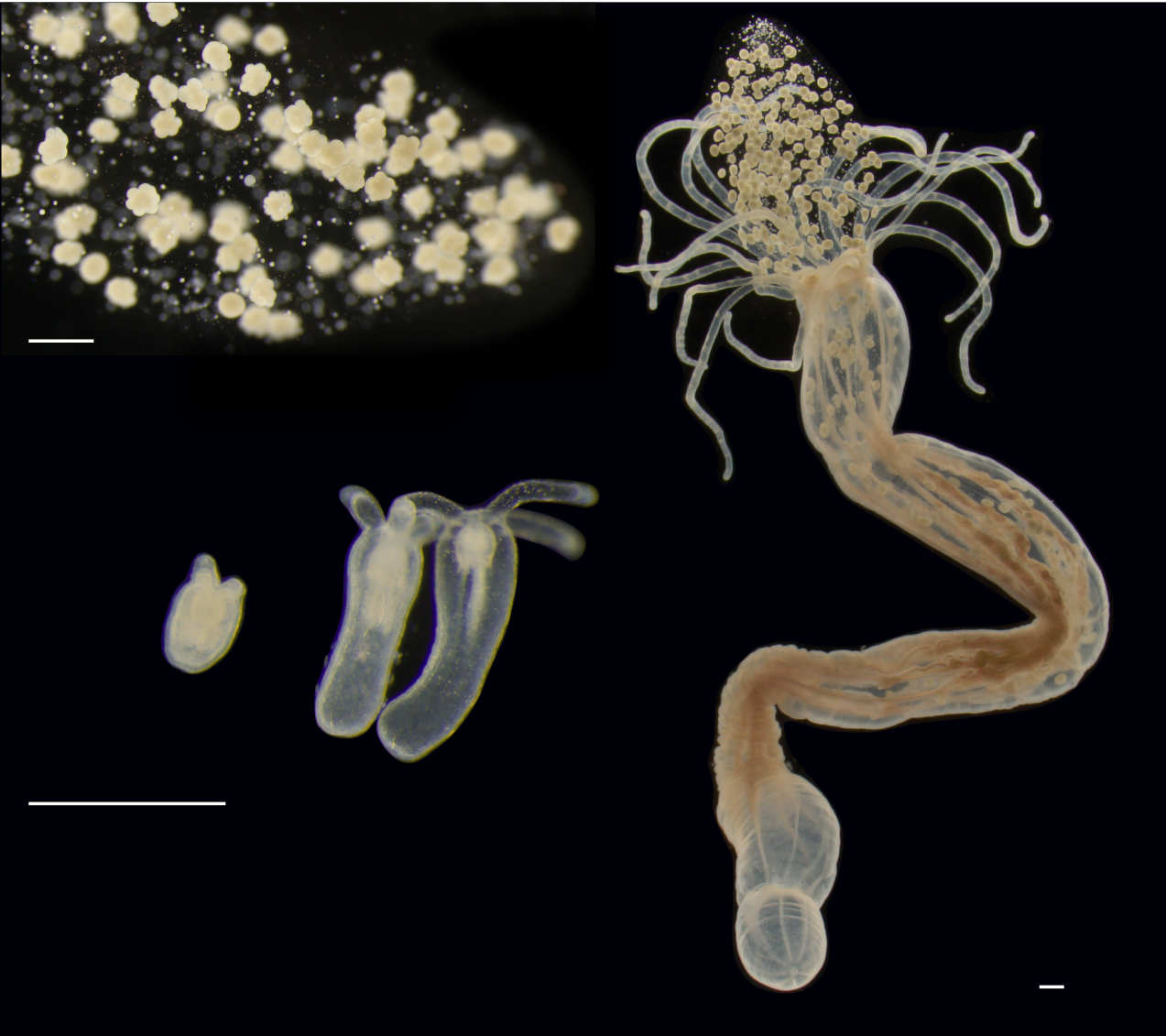 Various stages in the life of the sea anemone Nematostella vectenis. Top left: eggs, center left: various stages of growth, right: the adult animal. Image Credit: Heinrich Heine University Düsseldorf/Hanna Domin.
Various stages in the life of the sea anemone Nematostella vectenis. Top left: eggs, center left: various stages of growth, right: the adult animal. Image Credit: Heinrich Heine University Düsseldorf/Hanna Domin.
A team of researchers, led by Heinrich Heine University Düsseldorf (HHU) and involving scientists from Düsseldorf and Kiel, has employed the sea anemone Nematostella vectenis as a model to investigate the co-development of the microbiome with its host.
Published in the scientific journal Microbiome, the study discloses that in the early stages of life, the host organism predominantly governs the bacterial community, with interactions among bacteria taking precedence in subsequent development.
Across multicellular organisms, spanning from the simplest life forms to humans, a symbiotic relationship exists with a diverse array of microorganisms collectively referred to as the microbiome.
This microbiome comprises bacteria, fungi, viruses, and other components, serving various functions from metabolic processes to immune defense. For example, the human intestine relies on the microbiome to facilitate the absorption of nutrients from food, a critical aspect of the body’s well-being.
The central inquiry in this study revolves around the concurrent development of the microbiome and its host.
The researchers observed that the composition and proportion of microorganisms in the sea anemone Nematostella vectenis undergo significant changes throughout its life cycle, reaching a stable form only in adulthood.
The inquiry focused on understanding whether the host governs the colonization by specific microbes or if the microbes autonomously regulate themselves during the host’s maturation.
Addressing this question, a collaborative team from HHU, Kiel University (CAU), and the GEOMAR Helmholtz Centre for Ocean Research Kiel, conducted the study under the leadership of Professor Dr Sebastian Fraune from the Institute of Zoology and Organismic Interactions at HHU. The research was carried out within the framework of the Collaborative Research Centre (CRC) 1182 “Origin and Function of Metaorganisms,” overseen by CAU.
We took adult Nematostella polyps, which had no microbiome following intensive antibiotic treatment, and then recolonized them in a targeted way. To do this, we used bacterial communities that corresponded to those of firstly a Nematostella larva, secondly a juvenile animal, and thirdly an adult polyp."
Dr Hanna Domin, Study Lead Author, Heinrich Heine University Düsseldorf
The study has currently been published in the Microbiome journal.
In each of the three instances, the researchers investigated the chronological development of the microbiome.
Their findings revealed that only the primary colonizers, specifically the bacteria constituting the microbiome of the youngest animals, successfully established themselves in the adult polyps. In contrast, the bacteria from older animals faced challenges in establishing a foothold.
Following recolonization, the microbiome then undergoes a development process that is very similar to the normal development of host and microbiome. It takes around four weeks to reach the same status as adult animals that have undergone a normal growth process."
Dr Sebastian Fraune, Study Corresponding Author and Professor, Institute of Zoology and Organismic Interactions, Heinrich Heine University Düsseldorf
As a result of their observations, the researchers deduce that the host, likely through its intrinsic immune system, regulates the composition of the initial colony.
Domin noted, “However, the host no longer has a significant influence over the further development of the microbiome after this point. The bacteria control this themselves and lay suitable foundations for their descendants.”
A crucial element of the project, advanced by the research team led by Professor Dr Christoph Kaleta in Kiel, focused on scrutinizing metabolic networks. This entailed exploring the connections among various bacteria through their metabolism and understanding how they mutually influence each other.
“We were able to identify metabolic pathways, which are specific to the initial colonisers as well as pathways that only play a role at a later stage,” states Dr Johannes Zimmermann from CAU.
The research team determined that the breakdown of the polysaccharide chitin is pivotal, especially for the initial colonizers.
It was only recently revealed that Nematostella has the capability to produce chitin. However, the purpose of this chitin production remained unknown, as these animals, unlike insects, do not require chitin for their structural development.
Fraune added, Our results provide clear indications that chitin plays a role for the microbiome.”
While the sea anemone possesses solely an innate immune system, the findings hold significance for medical research. Newborns encounter numerous bacteria shortly after birth, and during this phase of their lives, they, too, rely solely on an innate immune system.
Therefore, the crucial aspect of the initial colonization with the right microbes is essential not only for establishing a functional microbiome but also for training the adaptive immune system in humans.
It has been noted that the microbiome development of children born by Caesarean section who have only limited contact with the bacterial communities of the mother during birth frequently differs to that of children born naturally."
Dr Sebastian Fraune, Study Corresponding Author and Professor, Institute of Zoology and Organismic Interactions, Heinrich Heine University Düsseldorf
However, disrupted microbial colonization during the early development phase changes metabolic and immune programming and appears to be connected with an increased risk of the immune system and metabolic disorders.
For this reason, initial studies are being conducted in which children born by Caesarean section are brought into contact with the vaginal secretions of the mother immediately after birth to ensure natural initial colonization.
Source:
Journal reference:
Domin, H., et al. (2023) Sequential host-bacteria and bacteria-bacteria interactions determine the microbiome establishment of Nematostella vectensis. Microbiome. doi.org/10.1186/s40168-023-01701-z.